colloque bobigny 2017 resultats-pivots
Colloque_2017_bobigny-poster-SSP_G_DELEPINE
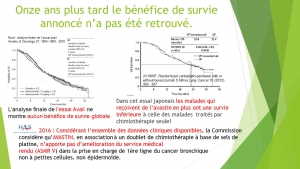
two presentations in french with curves of the results
“Are the results of pivotal studies in oncology reliable?”
by G. Delépine, N. Delépine, S. Alkhallaf
Introduction
Over the last fifteen years, the authorization to put innovative therapies on the market has been granted after one or sometimes two short placebo-controlled studies on a small number of patients. To see if these pivotal studies are reliable, we compared their initial results with the latest published results.
Material and methods
A computerized search with key words Avastin, herceptin, erbitux, erlotinib, sorafenib, gefitinib, crizotinib, afatinib, temsirolimus, pazopanib, sunitinib, axitinib, in bronchopulmonary squamous cell carcinoma, kidney, colon, breast and ENT cancers has been realized to find 42 pivotal studies. We then looked for those with distant results specifying the duration of progression-free survival, Overall survival and toxicity were published (32) and compared the results of the pivotal studies with those of the last trials on these three criteria.
Results
Less than 30% of the results presented to the regulatory agencies during marketing authorization applications are fully confirmed by subsequent trials. 20% of them are confirmed for 1 or 2 criteria. The discrepancies observed between initial and late results are always in the direction of greater efficacy or less toxicity of the new drug in the pivotal study.
Conclusions
In targeted therapies of solid tumors the favourable initial results of the pivotal studies are rarely confirmed by subsequent publications and the discrepancy still favours the new drug, suggesting that many of these trials do not represent real population use and that some of them benefited from the » improvement » of their results before presentation to regulatory agencies.
Conference « Overconsumption, overtreatment » of Medicine Faculty of Bobigny, Seine Saint Denis (France)
Summary of a communication (April 2017)
SEE THE POSTER http://surmedicalisation.fr/?page_id=4556#Gerard_DELEPINE1 IN FRENCH
